 
So, if all the coefficients can be divided by 2 or 3, do this before finalizing the reaction. A balanced equation is reduced to the lowest whole number coefficients.If there is only one mole or one atom, then the coefficient or subscript "1" is implied, but is not written.This is a requirement the equation must satisfy to be consistent with the law of conservation of matter. If the atom appears in more than one reactant or product, add together all the atoms on each side of the arrow. The chemical equation described in section 4.1 is balanced, meaning that equal numbers of atoms for each element involved in the reaction are represented on the reactant and product sides. There are no more or less atoms at the end of a chemical reaction than there were at the beginning. The law of conservation of mass states that matter cannot be created nor destroyed. To calculate the number of atoms, multiply the coefficient and the subscript. Li + H2O LiOH + H2 L i + H 2 O L i O H + H 2.Subscripts (numbers below an atom) indicate the number of atoms in a single molecule. Coefficients (number in front of a chemical formula) indicate moles of a compound.  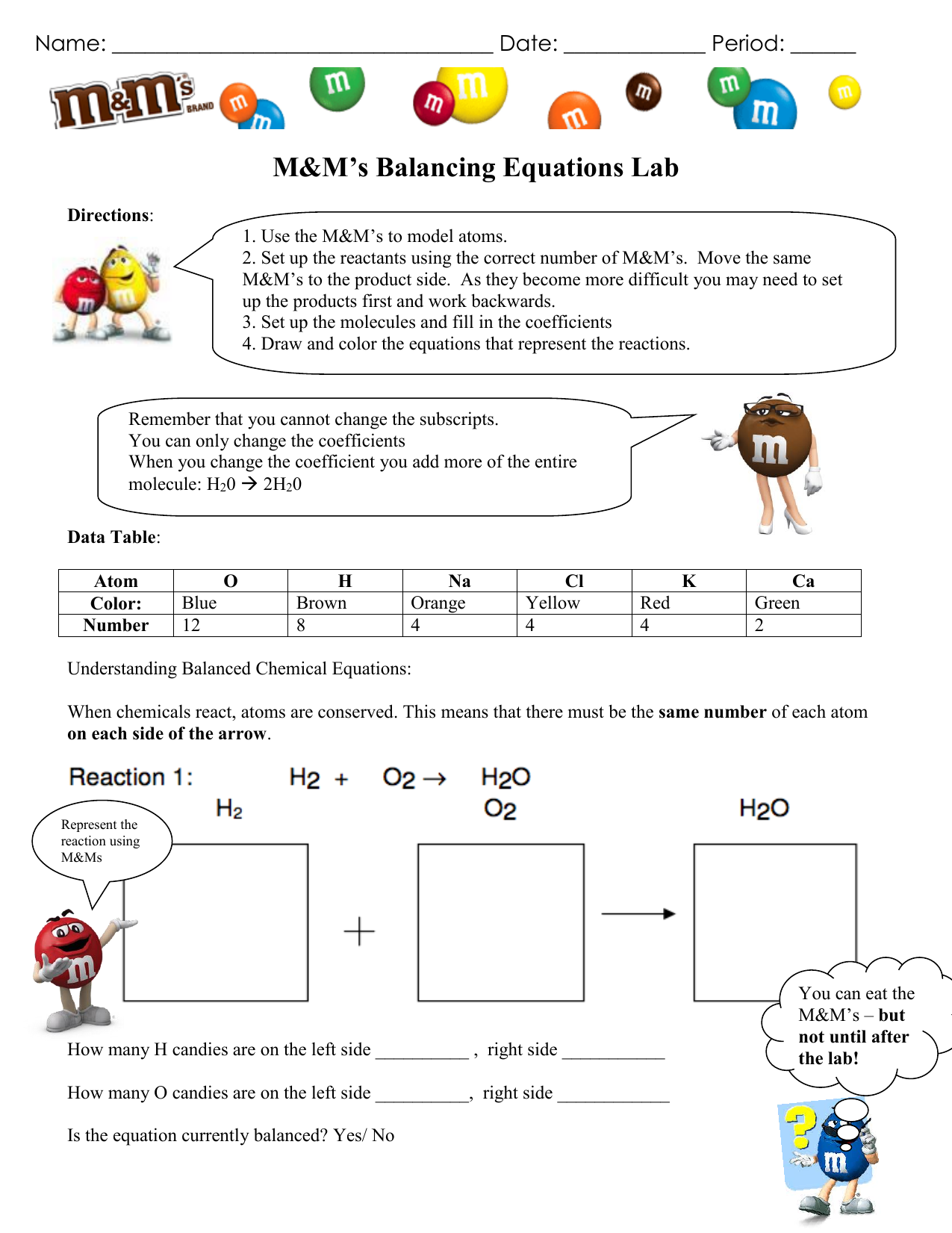
To write a balanced equation, the reactants go on the left side of the arrow, while the products go on the right side of the arrow.A balanced equation contains the same number of each type of atoms on both the left and right sides of the reaction arrow. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
